For perfectly crystalline substance entropy S = 0 at 0 KĪbsolute entropy of a pure substance : Absolute value of entropy (unlike absolute value of enthalpy) can be calculated at any temperature. Pure corresponds to no mixing of foriegn substances. 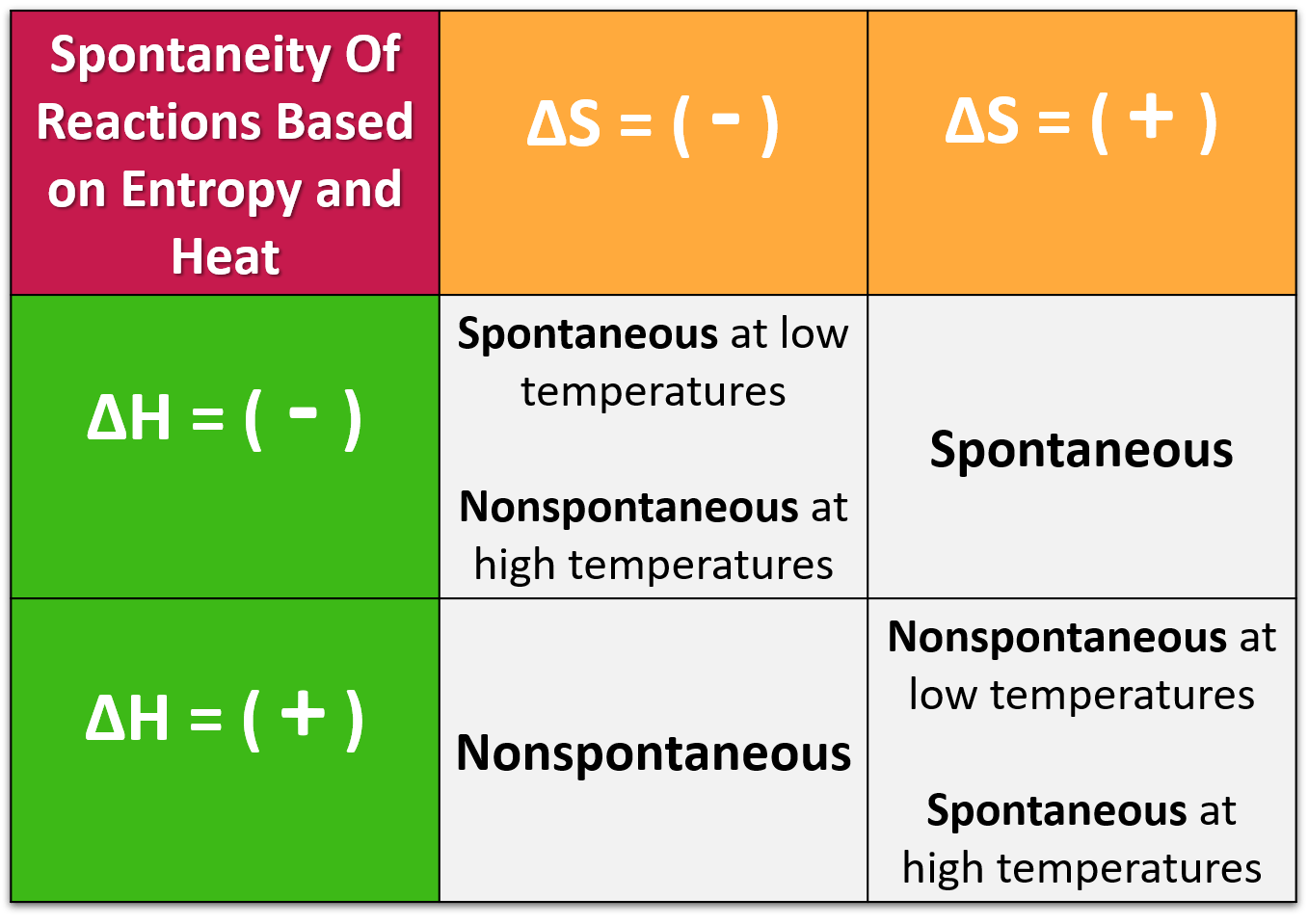
Kinetic energy is directly proportional to Temperature, when temperature becomes zero then kinetic energy will also be zero. It is also influenced by the enthalpy change (H) and the absolute temperature (T) of. There are a wide variety of crystallographic defects. However, the spontaneity of a reaction is not determined by entropy alone. A perfect crystal is a crystal that contains no point, line, or planar defects. Third law of thermodynamics : Entropy of pure and perfectly crystalline substance at 0 K is zero. JEE Main 2022 Question Paper Live DiscussionĪccording to third law of thermodynamics absolute value of entropy can be caculated at any temperatures. The entire architecture of thermodynamics is built on carefully defined terms, many of which have an everyday meaning that is not exactly what chemists mean.While much effort has been devoted to measuring and calculating the enthalpies of well-defined adsorbates, few measurements of the entropies of adsorbates have been reported. Difference Between Selling And Marketing Theoretical calculation of both the rate and equilibrium constants for such reactions requires knowing the entropy and enthalpy of the adsorbed molecule. 
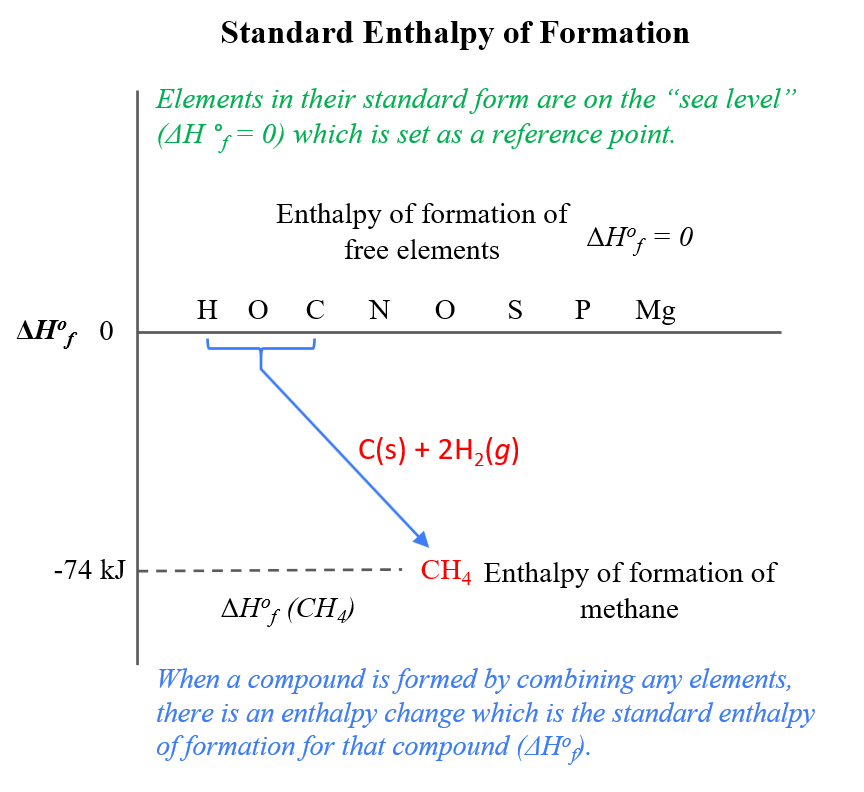
TS Grewal Solutions Class 11 Accountancy.TS Grewal Solutions Class 12 Accountancy.CBSE Previous Year Question Papers Class 12.CBSE Previous Year Question Papers Class 10.As we have seen with enthalpy, it was not really possible set (H(0)) to zero. (d) Calculate the entropy change, Scomb, for the reaction and account for the. NCERT Solutions For Class 6 Social Science It is tempting to set the (S(0)) to zero and make the entropy thus an absolute quantity. (c) Calculate the standard molar enthalpy of formation, Hf, of propane gas.NCERT Solutions for Class 7 Social Science.Now there you have them, the textbook entropies of ions, which are not quite absolute. It is kind of natural to consider its entropy 0 0, much like the absolute elevation of the sea level is 0 0. NCERT Solutions for Class 8 Social Science Why, lets say it is HX+ H X +, because so much of our chemistry is dominated by the acid/base equilibria. Since we now know the entropy at T 0 K and we can calculate the entropy change for a temperature and phase change, we can then calculate the absolute entropy.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
